Research
Overview
Our group builds new instruments to make unique measurements that have previously been out of reach. Using tools from atomic physics and quantum optics, we aim to impact diverse fields including fluid and statistical mechanics, acoustics, and medicine.
Fundamental Physics: Short-Time Brownian Motion
Since 2010, we have built new experiments to trap micron-sized glass beads in liquid, air, and vacuum. We have also built a novel detection system to monitor the real-time position of a trapped bead on very short time and length scales.

One motivation for this work was a long-standing prediction in physics: In 1907, Albert Einstein published a paper where he considered the instantaneous velocity of a Brownian particle and showed that it could be used to test the equipartition theorem, one of the basic tenets of statistical mechanics. However, Einstein concluded that it would be impossible to measure instantaneous velocity in practice due to the very rapid randomization of the motion.
We have measured the instantaneous velocity of a Brownian particle, over 100 years since Einstein's prediction. Our velocity data was used to verify the Maxwell-Boltzmann velocity distribution and the equipartition theorem for a Brownian particle. We have conducted similar measurements in liquid and resolved the instantaneous velocity. The physics in the case of liquid is much richer than air due to viscous fluid effects like memory and added mass.
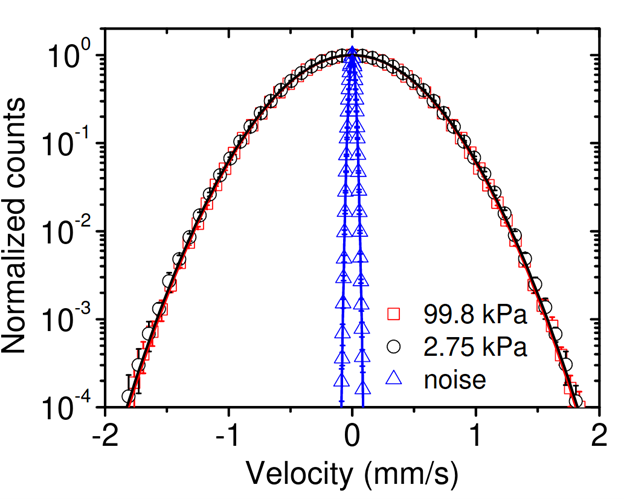
We have recently used the energy equipartition theorem to "weigh" a microscopic particle in air at the pico-gram level by fitting the mass to the observed Maxwell-Boltzmann distribution. Mass metrology at these levels has applications to single-particle aerosol science including cloud seeding and heterogeneous nucleation of super-cooled liquids, two phenomena that are, at present, poorly understood, but of general interest for climate science.
Also in air, we have demonstrated that optically trapped microbeads are sensitive high bandwidth acoustic transducers. Unlike traditional microphones, which measure acoustic pressure, the micro-bead is sensitive to acoustic velocity. Our micro-bead sensor has superior sensitivity to any commercially available device capable of directly measuring acoustic velocities. Because of the miniscule inertia of these micro-beads, the acoustic sensitivity at high frequencies is also superior to that of commercially available microphones, allowing our sensor to resolve the short rise times of acoustic impulses. Applications range from pinpointing the stopping point of protons in the body during proton therapy for cancer, to detection of acoustic events inside bubble chambers. The latter are being used to search for dark matter and cosmogenic neutrinos.
For Brownian motion in liquids, we are now developing a pulsed laser detection method that we believe will break into the experimentally new regime of compressible-flow Brownian motion. The compressible regime is characterized by time scales shorter than the speed of sound crossing the bead diameter. At these scales, added-mass effects break down and the bead's bare mass enters the equipartition theorem. Therefore, this system will enable the first precise test of the equipartition theorem for a colloidal particle in a classical liquid. We plan to use this system to study thermodynamics and statistical mechanics of small systems out of equilibrium and the emergence of the "arrow of time."
Medical Physics: Prevention of Iron Deficiency
Iron is an essential mineral for our health, needed for transporting oxygen to the brain and the body. Iron deficiency and the resulting anemia are widespread, affecting around two billion people worldwide, with a very high rate in children and in women during pregnancy due to the increased iron needs of these populations. It is considered a crisis in pediatric healthcare, due to the dire effect of iron deficiency on 50% of children worldwide and 15% of children in the US alone. Iron deficiency can cause severe developmental problems and permanent brain damage to infants and toddlers and decrease school performance in older children.

Although abundant in nature, iron is difficult to absorb from food, especially for young children where the need for the element is most critical and its lack potentially devastating and permanent. Even in affluent countries like the US, this problem is frequently undiagnosed and difficult to treat effectively.
Our collaborator and pediatrician Dr. Steven Abrams pioneered the use of completely safe, naturally occurring rare isotopes (iron-57 and iron-58) to assess and optimize absorption of iron into the body. The methodology is proven, but implementation with large populations has not been possible due to the prohibitive cost and low availability of the enriched isotopes, and the slow and tedious analysis of blood samples.
Quantum laser technology developed in our group has provided an unexpected solution to the isotope problem. Drawing on physics work manipulating individual atoms with laser trapping, we developed a way of controlling atoms to separate isotopes with high efficiency and extraordinary purity (over 99.95%). This method, termed Magnetically Activated and Guided Isotope Separation (MAGIS), can produce highly enriched isotopes of numerous elements at relatively low cost and high reliability.
We are exploring the use of fluorescence spectroscopy to detect stable isotopes of iron in blood. Previously developed dry-ashing techniques can be used to extract the iron content of blood samples as iron oxide powder. These iron oxide samples can be vaporized via laser ablation and collimated to produce beams of atomic iron suitable for isotopic spectroscopy. We then resolve the isotope peaks of naturally occurring iron-54, 56, 57, and 58 via fluorescence spectroscopy. Currently, we are working to improve the sample efficiency of our spectroscopic system and thus minimize the required blood volume to produce a diagnostic spectrum, which is an important criterion for applications involving infants and small children.

The work at UT will be further developed in conjunction with the non-profit Pointsman Foundation, where Mark Raizen is Founder and Chairman of the Board, www.pointsman.org. The goal is to translate scientific research into clinical practice. After iron, other essential mineral deficiencies such as zinc and calcium can be addressed with these techniques, to ensure the proper development of at-risk children. Possible future projects also include ultra-sensitive detection of toxic heavy metals, such as lead, cadmium, arsenic, and mercury, which can get into the water or food system and cause serious health problems, and determining exposure is of particular concern in children.
Link to previous research